Additif chimique 98 % d'engrais composés Phosphate monosodique - Chine Les produits chimiques, produits chimiques de traitement chimique de l'eau
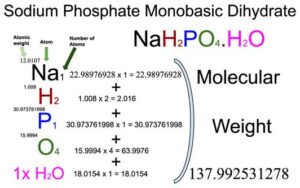
Sodium Phosphate Monobasic Monohydrate (NaH2PO4.H2O) Molecular Weight Calculation - Laboratory Notes

SOLVED: A buffer was made by mixing aqueous solutions of NaH2PO4 and Na2HPO4 together. This buffer is made by mixing two salts together. a. Write the balanced dissociation reaction for solid NaH2PO4

1 Bán Hóa chất Sodium dihydrogen phosphate monohydrat, reagent grade - NaH2PO4.H2O - SO0331 - Scharlau giá rẻ ở hcm

Welcome to Chem Zipper.com......: In a determination of P an aqueous solution of NaH2PO4 is treated with a mixture of Ammonium and magnesium ions to precipitate magnesium Ammonium phosphate (Mg(NH4)PO4.6H2O. This is
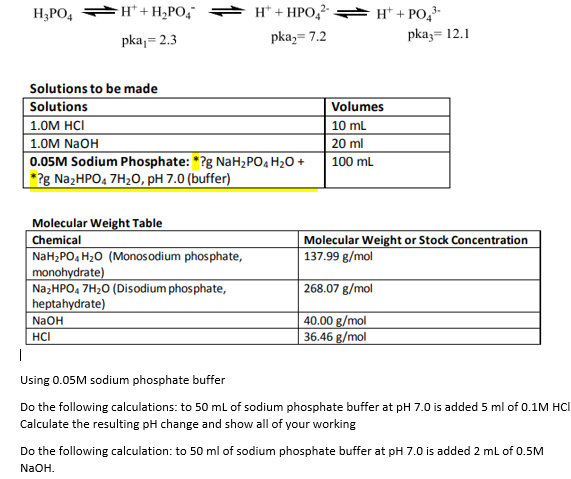
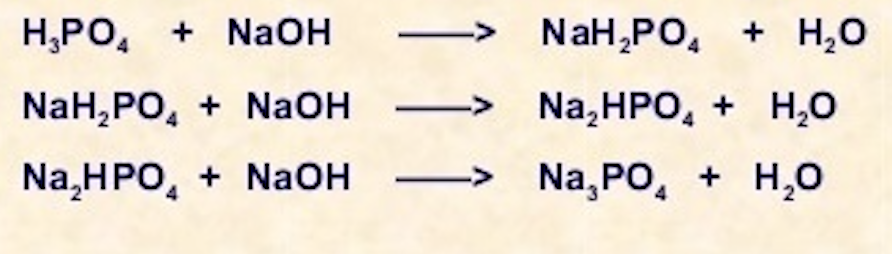
8. Calculate equivalent weight of H3PO4 and Ca(OH)2 on the basis of given reaction H3PO4 + NaOH = NaH2PO4 + H2O Ca(OH)2 + HCl = Ca(OH)Cl + H2O

ReasonH_3PO_4 is a tribasic acid.AssertionNaOH + H_3PO_4 rightarrow NaH_2PO_4 + H_2O In the given reaction, the equivalent weight of H_{3}PO_{4} is M/3.

Mono Sodium Phosphate Water Treatment Chemicals Nah2po4. H2O - China Monosodium Phosphate, Sodium Dihydrogen Phosphate | Made-in-China.com
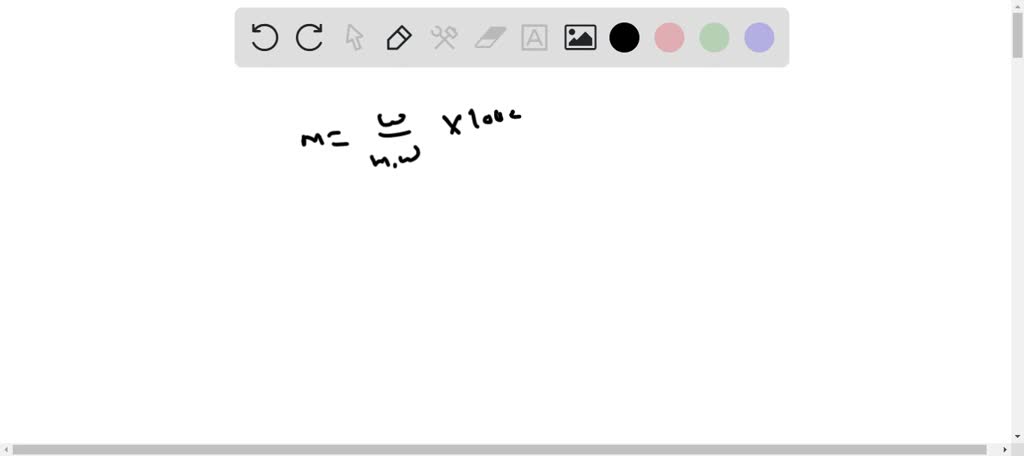
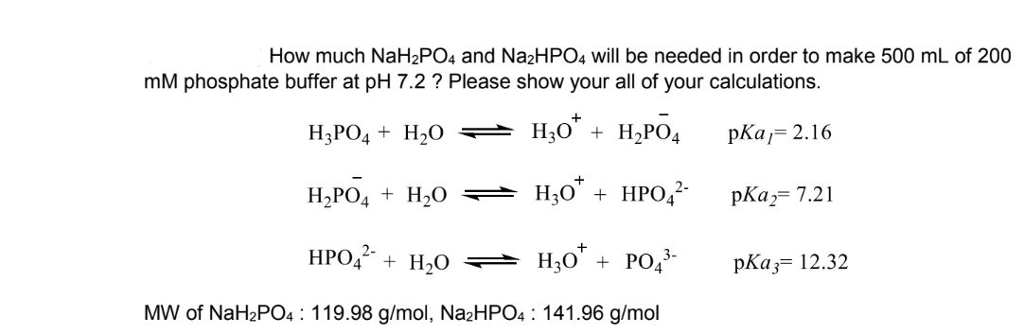
SOLVED: Weigh out 10.4 g sodium phosphate (NaH2PO4.H2O, MW = 138 g/mol). Dissolve the NaH2PO4.H2O in 450 mL water. Bring pH of solution to 7.4 with 10 M NaOH (need about 2
![Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 1kg) [CN04-1KG] - $40.00 : Bioland Scientific, for Your Research Needs Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 1kg) [CN04-1KG] - $40.00 : Bioland Scientific, for Your Research Needs](https://www.bioland-sci.com/images/NaH2PO4s%201KG.jpg)
Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 1kg) [CN04-1KG] - $40.00 : Bioland Scientific, for Your Research Needs