
Revision of the NaBO2–H2O phase diagram for optimized yield in the H2 generation through NaBH4 hydrolysis - ScienceDirect

Measurement of Specific Heat Capacity of NaBO2(aq) Solution and Thermodynamic Modeling of NaBO2 + H2O, NaBO2 + NaCl + H2O, and NaBO2 + Na2SO4 + H2O Systems | Journal of Chemical & Engineering Data

Diagram of physicochemical property versus sodium chloride content for... | Download Scientific Diagram

Phase diagram of the ternary system (LiBO2 + NaBO2 + H2O) at 288.15 K.... | Download Scientific Diagram

Phase Equilibria and Physicochemical Properties of the Ternary System (LiBO2 + NaBO2 + H2O) and Quaternary system (NaBO2 + NaCl + Na2SO4 + H2O) at 323.15 K | Russian Journal of Inorganic Chemistry
![PDF] Revision of the NaBO2–H2O phase diagram for optimized yield in the H2 generation through NaBH4 hydrolysis | Semantic Scholar PDF] Revision of the NaBO2–H2O phase diagram for optimized yield in the H2 generation through NaBH4 hydrolysis | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e8a022f49dd8cefe422671bdbf90195d1a22c0ee/32-Table2-1.png)
PDF] Revision of the NaBO2–H2O phase diagram for optimized yield in the H2 generation through NaBH4 hydrolysis | Semantic Scholar
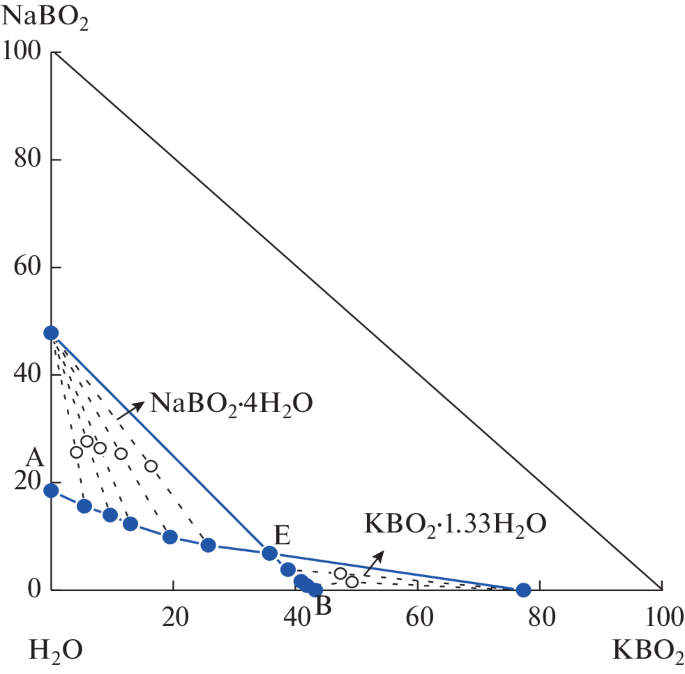
Solubilities, Densities, and Refractive Indices of the Quaternary System (LiBO2 + NaBO2 + KBO2 + H2O) and Subsystem (NaBO2 + KBO2 + H2O) at 288.15 K and 0.1 MPa | SpringerLink

Solid–Liquid Phase Equilibria in the Ternary Systems (LiBO2 + NaBO2 + H2O) and (LiBO2 + KBO2 + H2O) at 288.15 K and 0.1 MPa | Journal of Solution Chemistry

Measurement of Specific Heat Capacity of NaBO2(aq) Solution and Thermodynamic Modeling of NaBO2 + H2O, NaBO2 + NaCl + H2O, and NaBO2 + Na2SO4 + H2O Systems | Journal of Chemical & Engineering Data

Measurement of Specific Heat Capacity of NaBO2(aq) Solution and Thermodynamic Modeling of NaBO2 + H2O, NaBO2 + NaCl + H2O, and NaBO2 + Na2SO4 + H2O Systems | Journal of Chemical & Engineering Data

Solubilities, Densities, and Refractive Indices in the Ternary Systems (LiBO2 + NaBO2 + H2O) and (LiBO2 + KBO2 + H2O) at 298.15 K and 0.1 MPa | Journal of Chemical & Engineering Data
![PDF] Revision of the NaBO2–H2O phase diagram for optimized yield in the H2 generation through NaBH4 hydrolysis | Semantic Scholar PDF] Revision of the NaBO2–H2O phase diagram for optimized yield in the H2 generation through NaBH4 hydrolysis | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e8a022f49dd8cefe422671bdbf90195d1a22c0ee/33-Table3-1.png)
PDF] Revision of the NaBO2–H2O phase diagram for optimized yield in the H2 generation through NaBH4 hydrolysis | Semantic Scholar

Revision of the NaBO2–H2O phase diagram for optimized yield in the H2 generation through NaBH4 hydrolysis - ScienceDirect
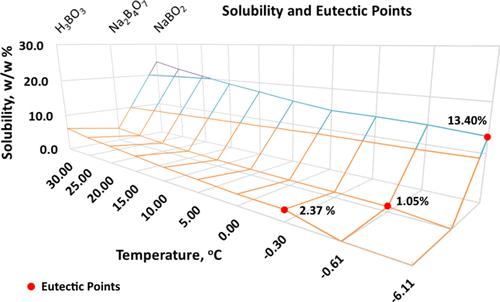
Eutectic Temperature, Density, and Solubility of H3BO3–H2O, Na2B4O7–H2O, and NaBO2–H2O Binary Systems,Journal of Chemical & Engineering Data - X-MOL

Solubilities, Densities, and Refractive Indices of the Ternary System (NaBO2 + KBO2 + H2O) at T = (298.15 and 323.15) K and P = 0.1 MPa | Journal of Chemical & Engineering Data









