Phase diagram of the H3BO3 + H2O system. Symbols represent experiemntal... | Download Scientific Diagram
Borazine (B3N3H6) hydrolyses slowly according to following reaction: B3N3H6 + H2O(excess)= NH3 + H3BO3 + H2. Weight of borazine required to form 0.2 mol H3BO3 is

Differences of water activity, aw, and enthalpy of dilution, ΔdilHm, of... | Download Scientific Diagram

Experimental determination and thermodynamic modeling of solid–liquid equilibria in the system NaCl–Na2SO4–H3BO3–H2O at 323.15 K and its application in industry - ScienceDirect

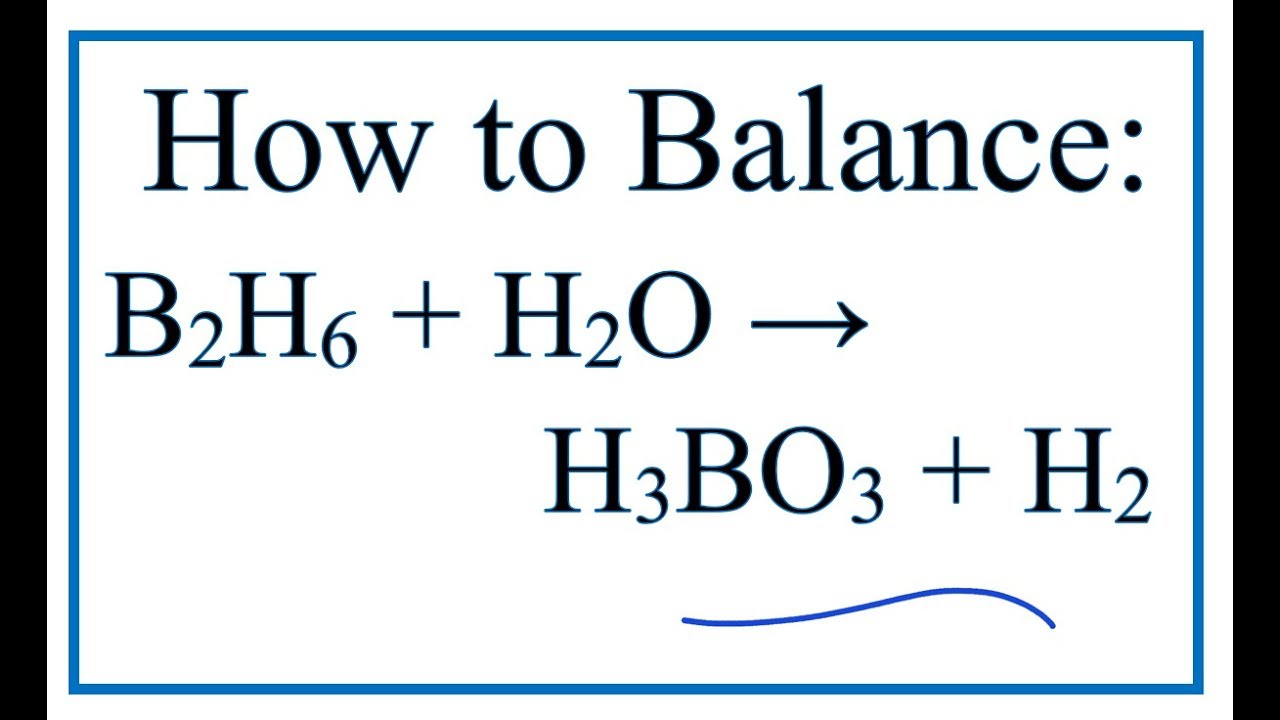
Be sure to answer all parts. Calculate the mass of each product formed when 70.76 g of diborane (B2H6) - brainly.com
![Write the reaction of action of heat on boric acid [H3BO3]? - Find 2 Answers & Solutions | LearnPick Resources Write the reaction of action of heat on boric acid [H3BO3]? - Find 2 Answers & Solutions | LearnPick Resources](https://www.learnpick.in/files/answerimages/5eb22de957ba09459f6fe1c23b3b4438.jpg)
Write the reaction of action of heat on boric acid [H3BO3]? - Find 2 Answers & Solutions | LearnPick Resources

Eutectic Temperature, Density, and Solubility of H3BO3–H2O, Na2B4O7–H2O, and NaBO2–H2O Binary Systems | Journal of Chemical & Engineering Data







![BORO 1,000 g/l [H3BO3 + H2O] ml 500 – Di Giovanni BORO 1,000 g/l [H3BO3 + H2O] ml 500 – Di Giovanni](https://www.digiovannisrl.it/cdn/shop/products/Titolchimica_2c125fb8-a963-4ba5-8e79-ee912fd88fc5_2048x2048.jpg?v=1643889520)